Answers
Answer:
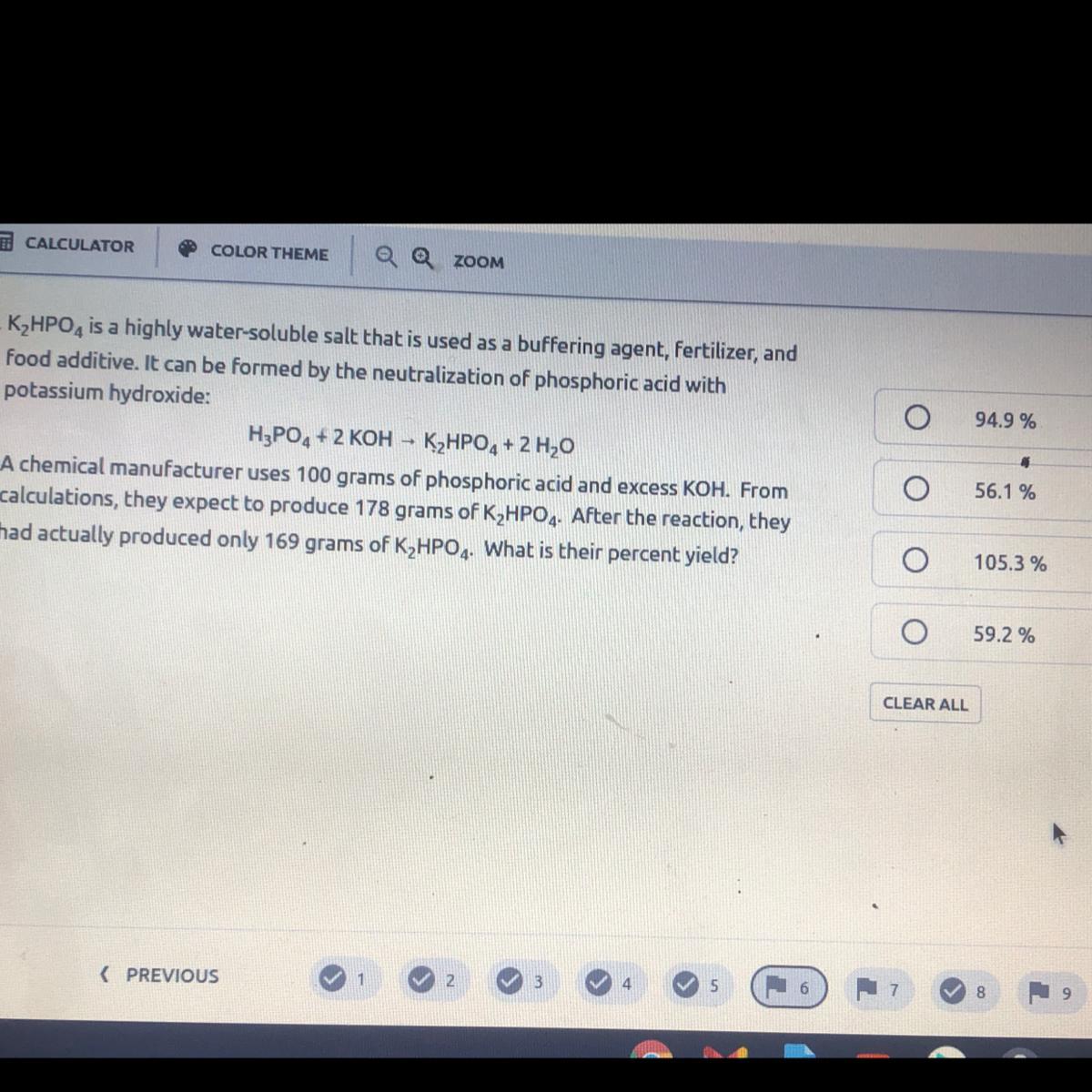
94.9% yield
Explanation:
Percent yield = actual yield/theoretical yield x 100
So you will divide the actual yield (what they really produced) over the theoretical yield (what they expected to produce). This will give you a decimal answer, which you multiply by 100 to get a percent.
169g/178g = 0.949 x 100 = 94.9%
Related Questions
You use a _____ when you are changing compounds
Answers
Answer:
coeffeicent!
Explanation:
this balances equations and also changes the amount of a compound/element present
Which two groups of the periodic table do not contain any non-metal?
Answers
The 17th and 18th i think.. If by non-metal you mean no metal I think Its the 17th and 18th
hope this could help a bit.
When mud and water are combined, the result is ______.
A. a mixture
B. an element
C. a molecule
D. a compound
Answers
Answer:
A. a mixture
Explanation:
hope this helps
You are mixing both mud and water so a mixture
Please help ASAP I’ll mark you as brainlister
Answers
Answer:
2
Explanation:
The balanced chemical equation for this reaction is:
2Na + K2S → 2K + Na2S
Meaning the coefficient for potassium (K) is 2.
Answer:
2 / 1 => 2 / 1
A beaker contains a sample of water with a mass of 325 grams. The water in the beaker
is boiled and weighed again after it cools. The new mass of the water is 305 grams. In
this example of the conservation of matter in physical changes,
A. the mass is less after it cools because water has less mass as a gas.
B. the mass of the water was 305 grams and the mass of the water vapor was 20 grams.
C. the process of boiling destroyed 20 grams of the water and left 305 grams of water.
D. the heat energy created by boiling water makes up the rest of the mass.
Answers
Answer: a
Explanation:
Bc I just did it and yeah
How many grams are in 1.5 moles of LiF?
solve for unknown
Answers
Answer:
The gram formula mass is the sum of the gram atomic masses of the atoms that make up a chemical formula. For lithium fluoride this works out to be 7g + 19g = 26g. Hence the mass of 1 mole of LiF is 26g; and the mass of 1.5 moles of LiF must be 1.5 × 26g = 39g.
Elizabeth collects different types of rocks whenever she goes on vacation. She has rocks that contain shiny crystals, grainy rocks, and rocks that are smooth. These rocks are formed? by different processes. Which of the following is NOT a process by which rocks are formed?
A. freezing of sediments.
B. compaction of sediments.
C. heat and pressure within the Earth
D. cooling of magma at Earth's surface
Answers
Answer:
It should be A. I've never heard of freezing of sediments
When one element replaces another element the reaction is known as a _ replacement reaction
Answers
Answer:
Single replacement reaction
Explanation:
A single replacement reaction, sometimes called a single displacement reaction, is a reaction in which one element is substituted for another element in a compound.
Which statement cannot be used in favor of solar energy
A) It does not produce any greenhouse gases.
B) There is a relatively high installation cost associated with solar panels.
C) New renewable jobs can be created with solar energy technology.
D) Solar energy from the sun is forever because the sun will exist forever.
Answers
4. A girl walked for 30 minutes. She noticed that she traveled farther in the first 15 minutes of
her walk than in the second 15 minutes,
What can she conclude about her walk?
A she walked over many hills
B
her average speed was faster during the first part of her walk
C. she walked in two different directions
D. she was walking at a constant speed
Answers
because her speed was faster in the first part of her walk
If you had 14.5 moles of gold, how many atoms would you expect to have?
Answers
Answer:
(14.5 mol) x (6.022 x 10^23 atoms/mol) = 8.73 × 10^24 atoms
Explanation:
pls mark brainliest
Convert the following problems:
a 96 kg to g
b 15 cL to kL
c 188 dL to L
d 3600 m to km
e 234 K to °C
f 43 °C to °F
i would really appreciate the help on this, I'm kinda struggling
Answers
Answer:
a.) 96000 g
b.) 0.00015 kL
c.) 18.8 L
d.) 36 KM
e.) -39.15 C
f.) 109.4 F
Explanation:
To turn kg to g, multiply the value by 1000
To Turn CL to KL, divide the volume value by 10000
To turn dl to l, divide volume by 10
to turn m to km, multiply meter value by 0.001 or divide by 1000
To convert c to f, multiply the temperature in degrees Celsius by 2, and then add 30
i hope this helps : )
Where are organelle located
Answers
Answer:
organelle are located in da cytoplasm of eukaryotic and prokaryotic cells
Hope this helps!
What is the mass in grams of 4.8 moles of Calcium Hydroxide?
Answers
Answer:
355.2 grams
Explanation:
m = M×n= (40 + 2(16+1))×4.8= 355.2 (g)
helpp
simplify the meaning of these
circuit
electric current
voltage difference
ohms law
resistance
Answers
Answer:
circuit
electric current
voltage difference
ohms law
resistance
Explanation:
Circuit - a roughly circular line, route, or movement that starts and finishes at the same place.
Electric current - A net unidirectional movement of electrons, or other charge carriers, caused by a potential difference
Voltage difference - A potential difference of one Volt is equal to one Joule of energy being used by one Coulomb of charge when it flows between two points in a circuit.
Ohms law -a law stating that electric current is proportional to voltage and inversely proportional to resistance.
Resistance - The refusal to accept or comply with something; the attempt to prevent something by action or argument.
If Hydrochloric Acid and Calcium Carbonate were combined would the reaction that occurs be endothermic or exothermic? Why?
Answers
Answer:
endothermic
Explanation:
more energy is being released and heat is being required to do the process.
Given 2Na + Cl2=2NaCl, what is the excess reactant? What is the limiting reactant?
Answers
Answer: The limiting reactant is Na
Explanation:
Which branch of chemistry would study how pollution effects marine life?
biochemistry
physical chemistry
materials chemistry
environmental chemistry
Answers
Answer: (D) environmental chemistry
Explanation:
The environmental chemistry is branch of chemistry would study how pollution effects marine life. option D is correct.
What is environmental chemistry?
Environmental chemistry is the study of the biochemical and chemical processes that occur in natural places like in forest, water bodies in air in soil and related to animals too.
Environmental chemistry is wide range of studies than air, water, soil, and chemicals. This also gives the brief account about the marine life with studying the life cycle of marine animals and plants too.
Therefore, environmental chemistry is branch of chemistry would study how pollution effects marine life. option D is correct.
Learn more about environmental chemistry, here:
https://brainly.com/question/25046469
#SPJ2
How many grams of chlorine are required in order to consume 8 mol of
sodium?
Answers
Explain why a climax community is not always a forest.
Answers
All changes in phase (solid-liquid-gas) are
Answers
Answer:
States of matter
Explanation:
Three states of matter
When a strontium atom loses two electrons to form an Sr2+ ion, the electrons are lost from the
a. 5s orbital. c. 3d orbital.
b. 5p orbital. d. 4f orbital.
Answers
Answer:When a strontium atom loses two electrons, it becomes a(n) cation with a charge of 2+. when any neutral atom loses an electron it becomes cation that is positively charged ions and an ion gets the charge according to the number of electrons loses.
you are very welcome but I may be inaccurate.
2. How will the equilibrium shift if the following changes are made? State if the reaction will shift
to the left or right and explain why it will shift in that direction.
PCl5 () + heat ⇌ PCl3 () + Cl2 ()
a. Temperature is increased
b. pressure is increased
Cl2 is added
d. PCl3 is removed as it is formed
3. How will the equilibrium shift if the following changes are made? State if the reaction will shift
to the left or right and explain why it will shift in that direction. (10 points each. 5 points for
correct answer, 5 points for correct explanation)
H2 () + Cl2 () ⇌ 2HCl() + heat
a. Heat is removed
b.Cl2 is added
d. PCl3 is removed as it is formed
3. How will the equilibrium shift if the following changes are made? State if the reaction will shift
to the left or right and explain why it will shift in that direction.
H2 () + Cl2 () ⇌ 2HCl() + heat
a. Heat is removed
b.HCl is added
c. H2 is added
d. Pressure is decreased
Use the reaction below to answer the following questions.
2NO2 ()⇌N2O4 ()
a. What is the expression for the equilibrium constant, Keq, for the reaction?
b. If there are 0.400 moles of N2O4 and 0.500 moles of NO2 in a 1.00 L flask at equilibrium at 20°C, what is the Keq?
Answers
Please give me brainleist. :)
Answer:
2a. If the temperature is increased, the reaction will shift to the right in an attempt to release some of the heat. As the forward reaction loses heat while the reverse would create more heat.
2b. If the pressure is increased, it would shift to the left to counteract the increase in pressure as the left side will have fewer molecules.
2c. If Cl2 is added the reaction will shift to the left in order to remove the stress of the extra Cl2 and favor the production of more reactant.
2d. If PCl3 is removed, the reaction will shift to the right. When part of the equation is removed the reaction learns to adapt to the loss by trying to make more Pcl3 and counteract the effects of losing the PCl3.
3a. The reaction will shift to the right to produce more heat and counter the negative effects of losing the heat.
3b. It will shift to the left to get rid of the excess HCl being produced and form more reactant from the breakdown of the HCl.
3c. It would shift to the right in order to get rid of the excess form products from it.
3d. If pressure is decreased there will be no effect on the shift of the reaction because there is an even amount of moles of gas on each side.
4a. K=[N2O4(g0] / [NO2(g)]2
4b. (Below)
K=[N2O4(g)] / [NO2(g)]2
0.4 / 0.5(2)
0.4/0.25 = 1.6
Keq= 1.6
The nucleus of an atom is ______ charged
Answers
Answer:
The nucleus of an atom is positively charged.
Explanation:
The nucleus of an atom is positively charged because :
The nucleus is a small dense region at the center of the atom. It consists of a positive protons and neutral neutrons. so, It has an overall positive charged.
Help on 2 and 3 due tomorrow pls help
Answers
Answer:
2
b= they are grouped differently, but all the atoms are still there.
What are sum things yhu can do to prevent brain damamge
Answers
Answer:
not bang your head
Explanation:
Answer:
focus on your surroundings
what system is the trachea is part of
Answers
respiratory tract
The trachea is part of the lower respiratory tract, along with the lungs, bronchi, bronchioles, and alveoli.
what is the mass of [tex]Fe^{2+}[/tex] in 5 tablets of iron if the number of moles of [tex]Fe^{2+}[/tex] is 4.2225 x [tex]10^{-4}[/tex] mol.
[ Ar = Fe, 56 ]
Answers
Answer:
Approximately [tex]0.11823\; \rm g[/tex] in total for the five tablets.
(Approximately [tex]2.3646 \times 10^{-2}\; \rm g[/tex] per tablet.)
Explanation:
The relative atomic mass of an element is the ratio between:
the mass of one atom of this element, and the mass of one-twelfth of a carbon-[tex]12[/tex] atom.The definition of moles ensures that the relative atomic mass of an element is approximately numerically equal to the mass (measured in grams) of one mole of the atoms of this element.
The question states that the relative atomic mass [tex]A_{\rm r}[/tex] of iron is [tex]56[/tex]. In other words, the mass of one mole of iron [tex]\rm Fe[/tex] atoms would be approximately [tex]56\; \rm g[/tex].
The question is asking for the mass of some amount of [tex]\rm Fe^{2+}[/tex] ions. Each [tex]\rm Fe^{2+}\![/tex] ion contains two fewer electrons than a neutral [tex]\rm Fe[/tex] atom. Hence, [tex]4.2225 \times 10^{4}\; \rm mol[/tex] of [tex]\rm Fe^{2+} \![/tex] ions might be lighter than the same number of [tex]\rm Fe \![/tex] atoms by a very small extent: The mass of one mole of electrons is approximately [tex]5 \times 10^{-4}\; \rm g[/tex], much smaller than the mass of the same number of [tex]\!\rm Fe[/tex] atoms (approximately [tex]56\; \rm g[/tex].)
Estimate the mass of these [tex]4.2225 \times 10^{4}\; \rm mol[/tex] of [tex]\rm Fe^{2+}[/tex] ions using the mass of the same number of [tex]\rm Fe[/tex] atoms:
[tex]\begin{aligned}&m({\rm Fe^{2+}}) \\ &\approx m({\rm Fe}) \\ &\approx 4.2225 \times 10^{-4}\; \rm mol \\ &\quad\quad \times 56\; \rm g \cdot mol^{-1} \\ &\approx 2.3646 \times 10^{-2}\; \rm g \end{aligned}[/tex].
Five of these tablets would contain approximately [tex]5 \times 2.3646 \times 10^{-2}\; \rm g \approx 0.11823\; \rm g[/tex] of [tex]\rm Fe^{2+}[/tex] ions.
3)
:NEN:
How many nonbonding electrons are on each nitrogen atom?
A)
0
B)
2
c)
4
D)
6
4)
-))
Answers
Answer:
your answer is C
Explanation:
The electron-dot structure of NH3 places one set of nonbonding electrons in the valence shell of the nitrogen atom. that means there are three bonded atoms and one single pair for a coordination number of four surrounding the nitrogen, the same occurs in water to. along with the Lewis dot structure for ammonia, NH3. the non bonding atoms are basically like hydrogen atoms the try to refuse to bond and they can't be fused unless the right conditions are met.
What is the molar mass of a sample of gas that has a density of 2.85g/L at 101kPa pressure
and 29°C? Name the law that is used to solve this problem.
Answers
Answer:71.25
Explanation:
The molar mass of a sample of gas that has a density of 2.85g/L at 101 kPa pressure and 29°C is 71.37 g mol⁻¹ and It can be solved using Ideal gas law.
What is Ideal gas law ?The ideal gas law is an equation of state that describes ideal gases.
This equation of state relates a gas’s pressure, volume, temperature, and mass, and is very useful for describing how gases will behave in ideal conditions.
This is the most common equation of state for gases.
Ideal Gas equation ;
PV = nRT
Molar mass (Mm) = m /n
Where ;
m = given massn = number of moleswe also know,
Density (D) = m / v
Where ;
m = massv = volumeNow, Putting the formula of Molar mass and Density in ideal gas equation we get ;
Molar mass = D RT/ P
Given ;
Density = 2.85 g/LPressure = 101kpa (= 0.99 atm)Temperature = 29°C (= 302.15 K)(Gas constant) R = 0.0821Putting all the values in the above equation of molar mass ;
Molar mass = 2.85 x 0.0821 x 302 / 0.99
= 71.37 g mol⁻¹
Learn more about Ideal gas law here ;
https://brainly.com/question/13821925
#SPJ2